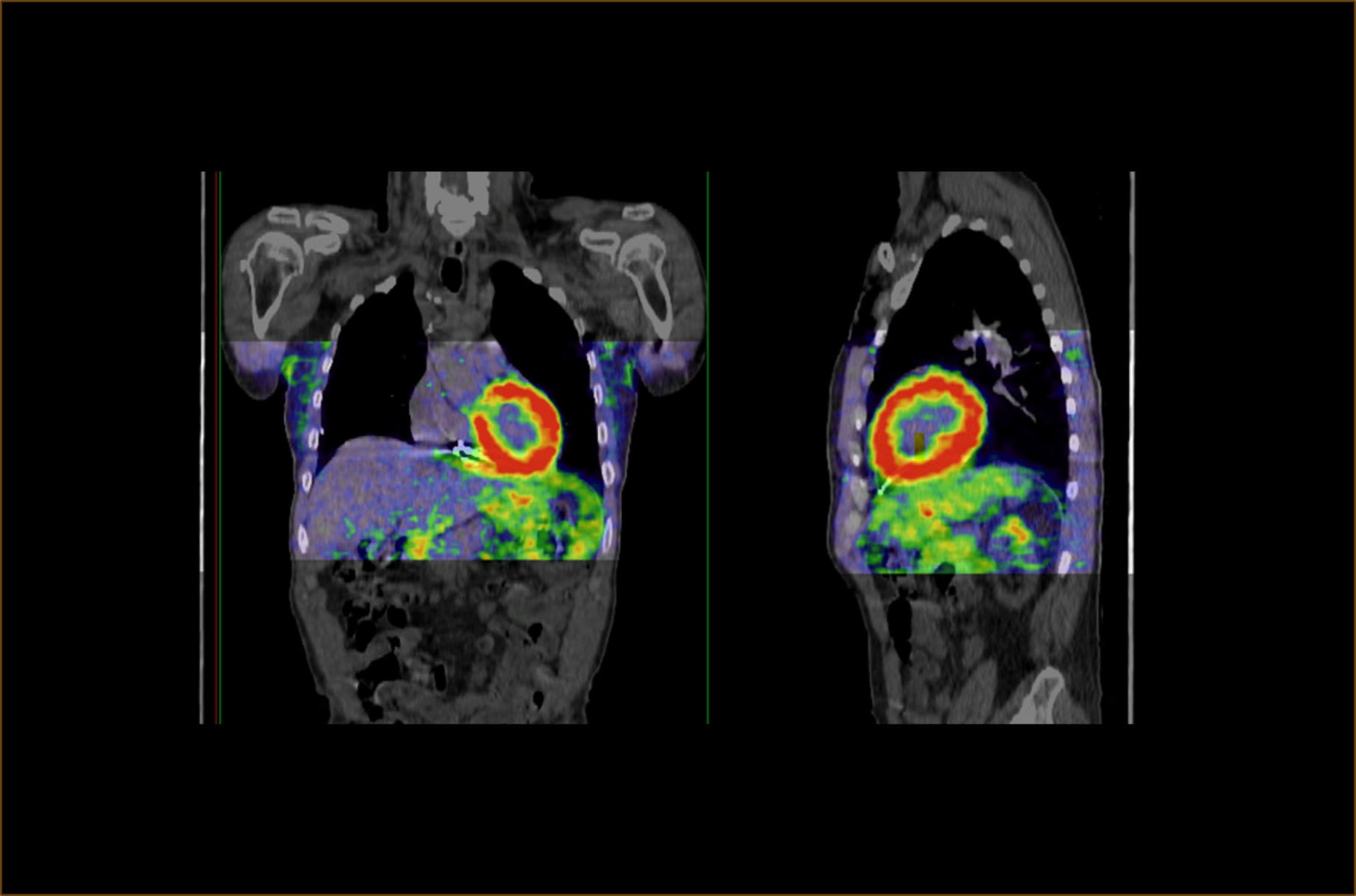
The Phase III REVEAL study met its primary endpoints demonstrating sensitivity and specificity of the PET/CT investigational radiotracer I-124 evuzamitide for diagnosing cardiac amyloidosis based on visual scan interpretation, according to a press release disseminated last week by Bayer.
I-124 evuzamitide PET/CT scan
(Source: press release)
The company said the complete results will be presented at an upcoming scientific conference and that it has “plans to discuss the data and submission for regulatory approval with the FDA and other health authorities.”
The role of PET in cardiac amyloidosis, including the potential role of I-124 evuzamitide, will be among topics discussed in the ICNC2026 session Imaging Cardiac Amyloidosis: State of the Art. The session will be co-chaired by ASNC Past President Sharmila Dorbala, MD, MPH, MASNC, who is also principal investigator of the REVEAL trial. For background on I-124 evuzamitide and other pan-amyloid radiotracers, check out this ASNC interview with Jonathan S. Wall, PhD, head of the team who invented the peptides on which the radiotracer was built.
Article Type
News & Announcements
Category
Publications, Research, Technologist, Trainee
Related Posts
ASNC, EANM Announce Winners of First Avijit Lahiri Young Investigator Awards Competition
ICNC2026 hosted the first Avijit Lahiri Young Investigator Awards Competition, where 6…
New JNC Compendium Offers Comprehensive Update on Cardiac Amyloidosis
The Journal of Nuclear Cardiology has just released a new JNC Compendium…
Dr. Mouaz Al-Mallah Will Present the Mario Verani Memorial Lecture at ASNC2026 in Las Vegas
Dr. Mouaz Al-Mallah Will Present the Mario Verani Memorial Lecture at ASNC2026…