ASNC is your source for the latest information emerging from the field of nuclear cardiology and cardiac imaging.
From our flagship scientific journal to our weekly newsletter, ASNC is dedicated to delivering high-quality research, news, and updates from the field.
ASNC News
The ASNC media team is available to arrange interviews with experts on the news featured in ASNC’s press releases, on research published in the Journal of Nuclear Cardiology, and on other developments in nuclear cardiology.
-

Cardiac PET with Myocardial Blood Flow Is Now the Preferred Test for Evaluating Coronary Artery Disease in All Patients
January 26, 2026 -

Jamieson M. Bourque, MD, MHS, FASNC, Installed as 2026 President of the American Society of Nuclear Cardiology
January 6, 2026 -

Dr. Juhani Knuuti to Deliver Keynote Lecture at ICNC2026, the International Conference on Nuclear Cardiology
November 24, 2025 -
New International Guideline Provides Expert Guidance for F-18 Flurpiridaz PET Imaging in Coronary Artery Disease
September 19, 2025

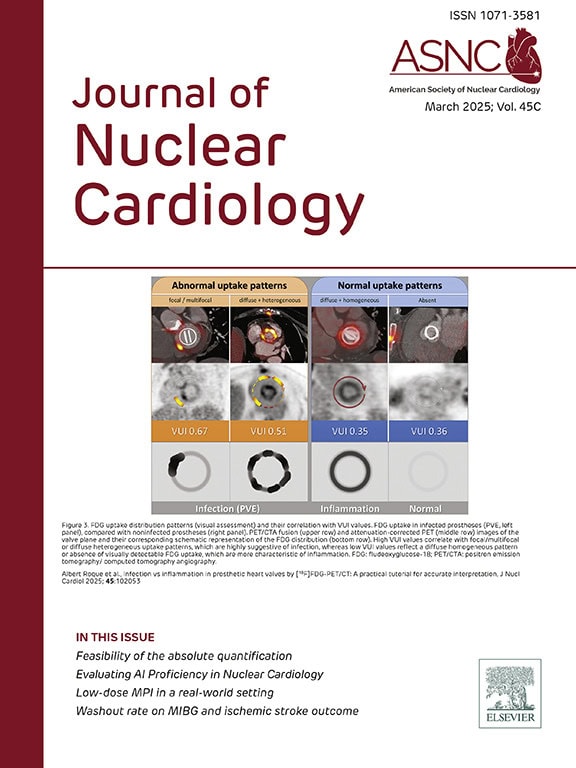
Journal of Nuclear Cardiology
The Journal of Nuclear Cardiology (JNC) is the only journal in the world devoted to this dynamic and growing subspecialty. The JNC is the official peer-reviewed publication of ASNC featuring the latest research findings and clinical developments in nuclear cardiology and related fields.
Latest Journal Issue
Read the latest issue of the Journal of Nuclear Cardiology (JNC). Full access to the JNC is available with an ASNC Membership. Join or renew today!
New JNC Podcast
The JNC CardioConnect podcast links experts, insights, and innovations in nuclear cardiology. Join Editor-in-Chief Marcelo F. Di Carli, MD, MASNC, and authors as they discuss important articles from the Journal of Nuclear Cardiology (JNC).

ASNC members receive the Society’s weekly Flashpoint newsletter that includes training and credentialing news, legislative and regulatory updates, international news, meeting and program information, and more.
ASNC Updates
Access the latest news and updates from ASNC. Search by category of interest, date, and more.
New Podcast! Is PET the Key to Finally Getting Heart Disease Diagnoses Right for Women?
Episode 13 of JNC CardioConnect highlights the results of the landmark AURORA…
Radiopharmaceuticals Expert Joins Lineup for ASNC’s May 5 Congressional Briefing on Improving Access to Cardiac Imaging
Etienne Montagut, president and CEO of PharmaLogic, will present at ASNC’s upcoming…
New Record! ASNC2026 Abstract/Case Submissions Achieve 30% Growth Over 5 Years
“This is an exciting time for all of us in nuclear cardiology,…
Receive ASNC SmartBrief
Sign up to receive ASNC's bi-weekly round-up of news and information related to nuclear cardiology and cardiovascular imaging.
ASNC members receive the Society’s weekly Flashpoint newsletter that includes training and credentialing news, legislative and regulatory updates, international news, meeting and program information, and more.

ASNC Interview Series
Explore the latest topics in nuclear cardiology through video conversations with luminaries and key leaders in the field.
ASNC Imaging Insights
Imaging Insights is a feature publication with in-depth articles that provide a deeper look at important developments and news in nuclear cardiology.

Advertise with ASNC
ASNC is where your marketing investment reaches the right audience and achieves optimal return.